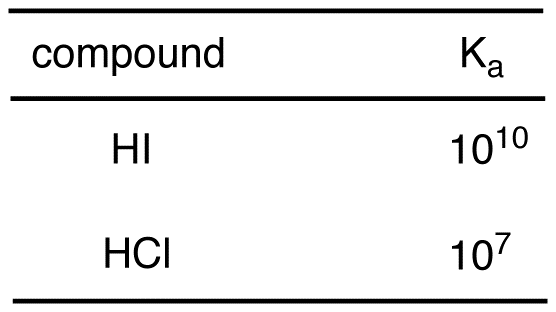
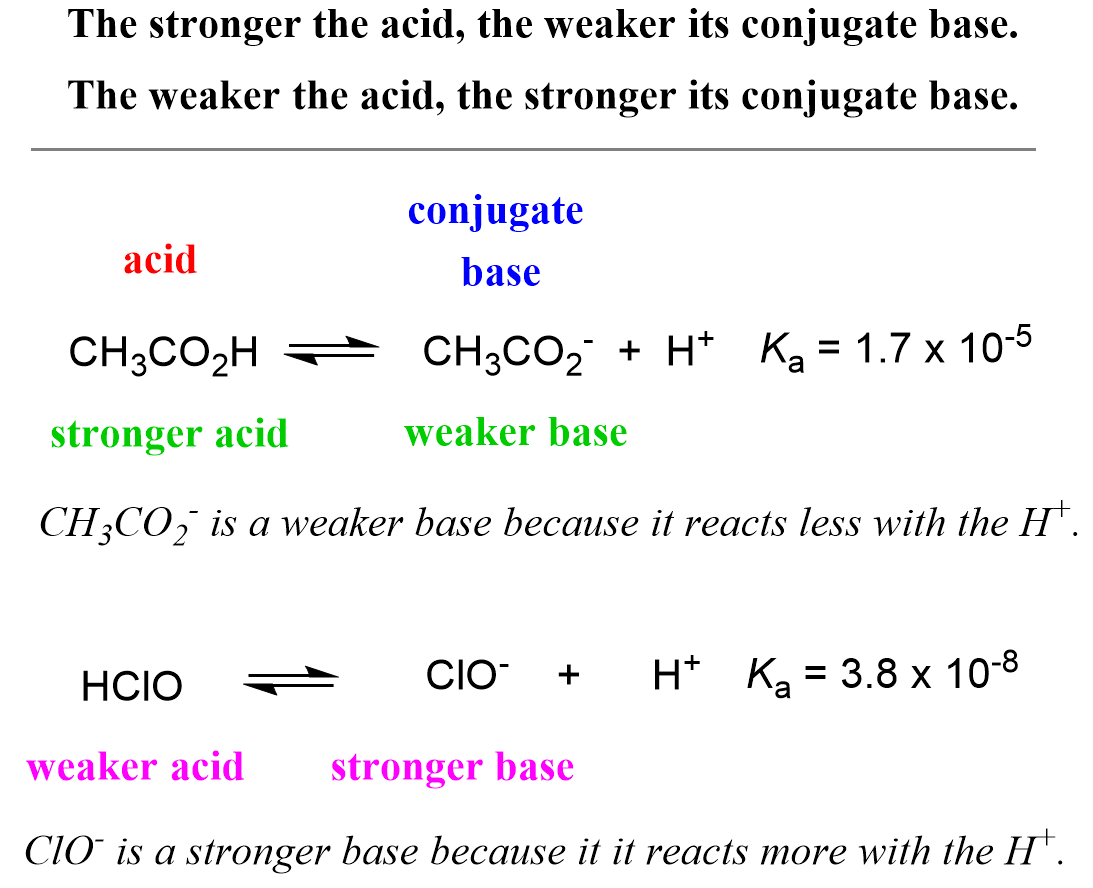
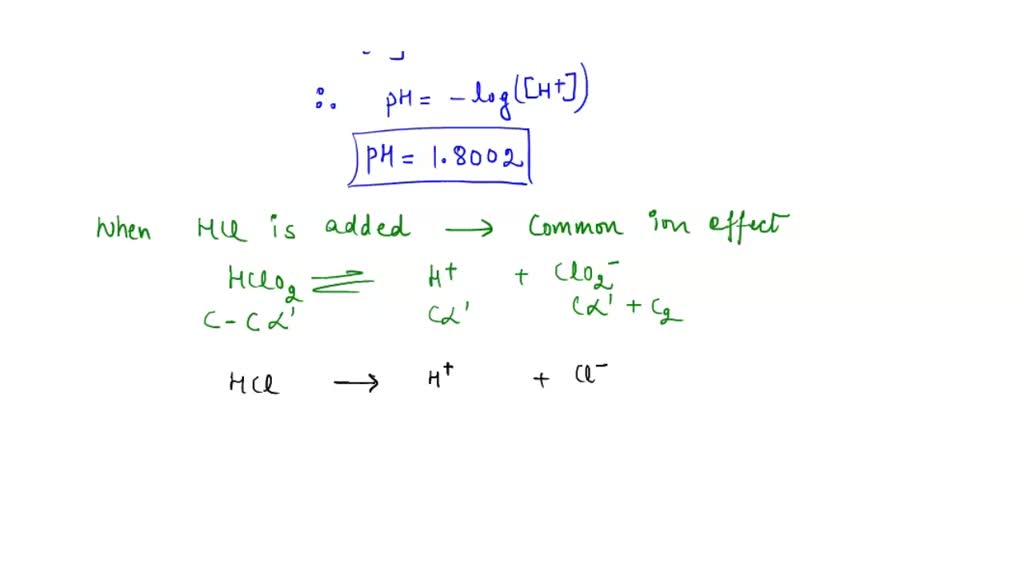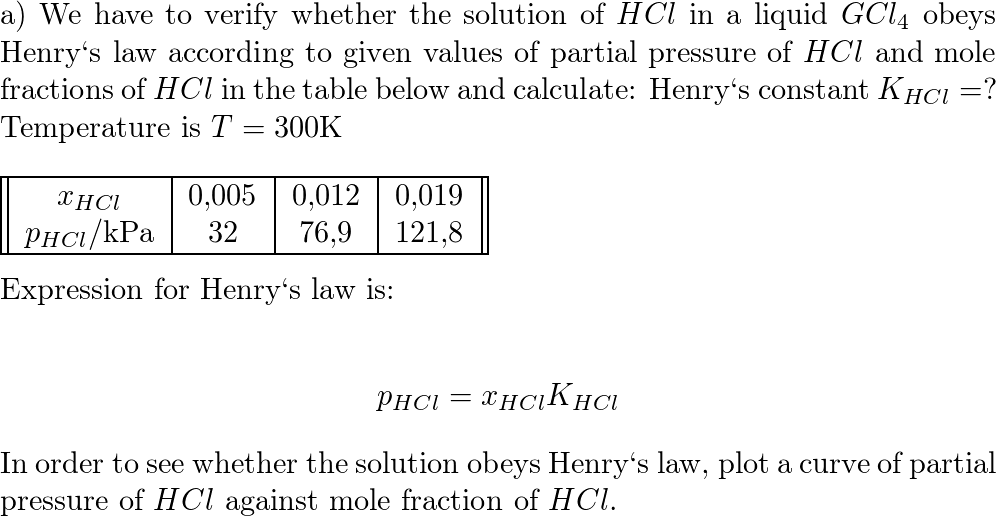An aqueous solution of a metal bromide MBr2 (0.05M) is saturated with H2S . The minimum pH at which MS will precipitate is X ? Ksp for MS = 6.0 × 10^-21 . [

Binding Association Constants (Ka ) and Binding Sites (N) for Three... | Download Scientific Diagram

The heat of formaiton of HCl at 348 K from the given data will be :1/2 H 2 g +1/2 Cl 2 g → HCl g H 2980= 22060 cal mol 1The

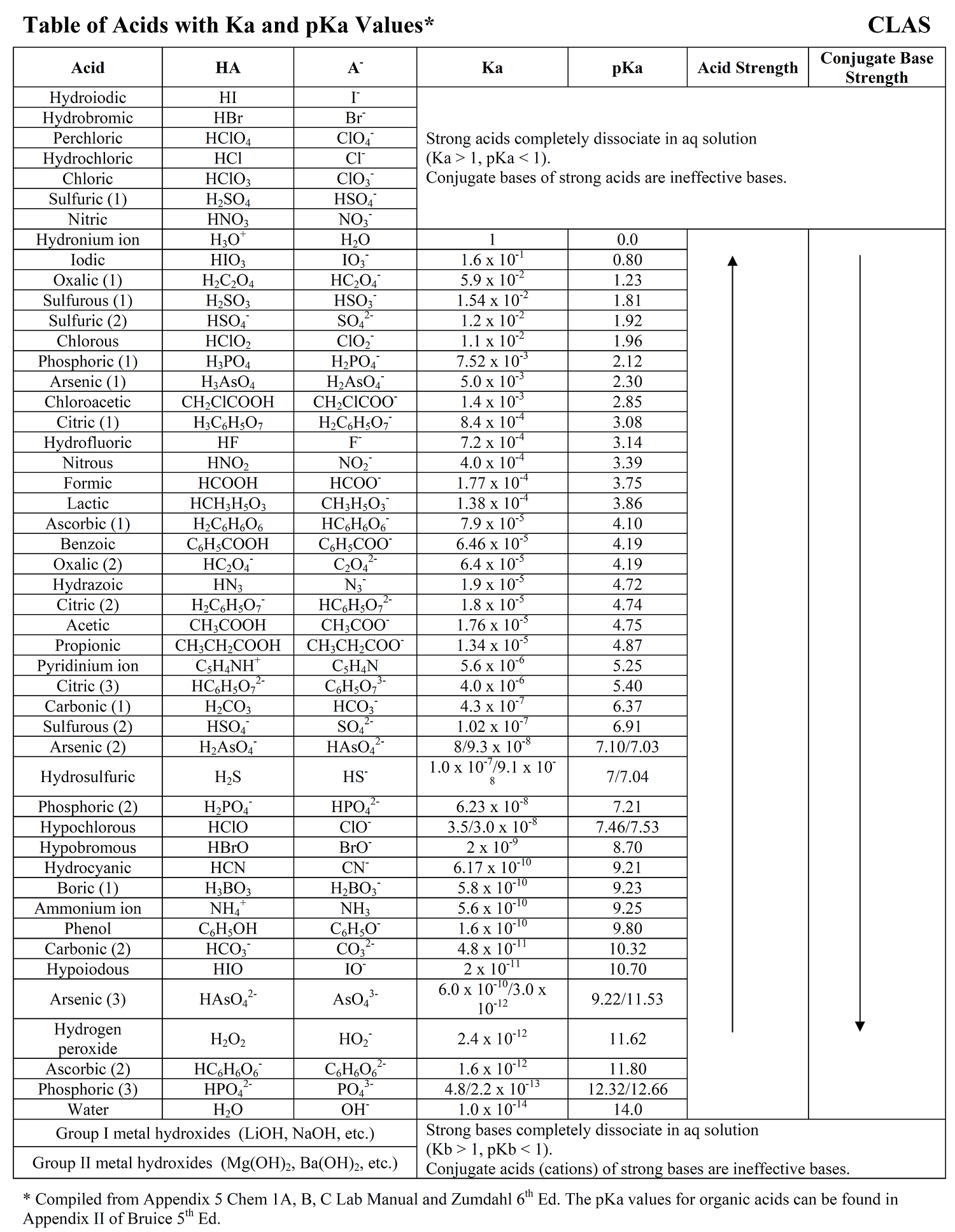
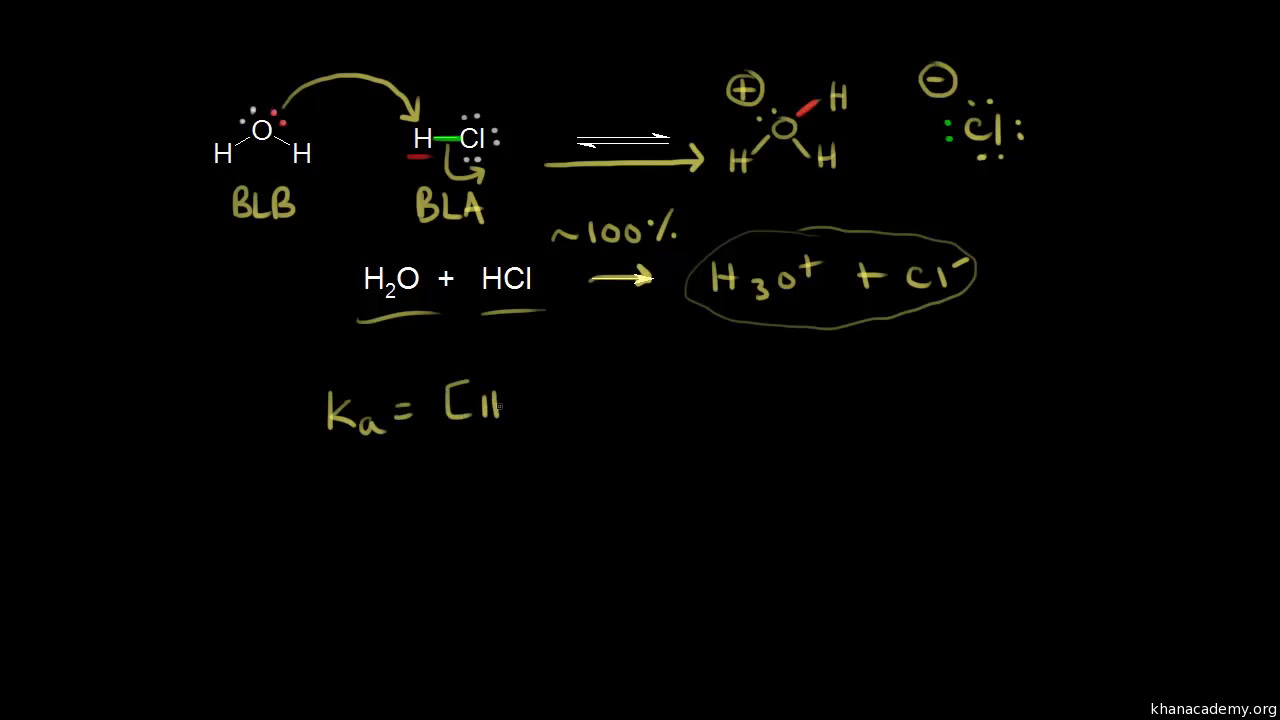
Ka for CH3COOH is 1.8×10^-5. find out the % dissociation of 0.2M CH3COOH in 0.1M HCl solution? - EduRev NEET Question

Reaction Kinetics of HCl Catalytic Oxidation over a Supported Cu-Based Composite Industrial Catalyst | Industrial & Engineering Chemistry Research

Calculate the work done (J) when 1 mole of zinc dissolves in hydrochloric acid at 273 K in a closed beaker at 300 K [Volume at STP = 22.4 L, rounded up